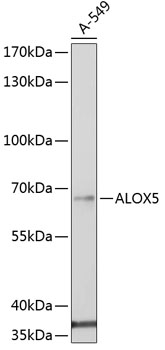
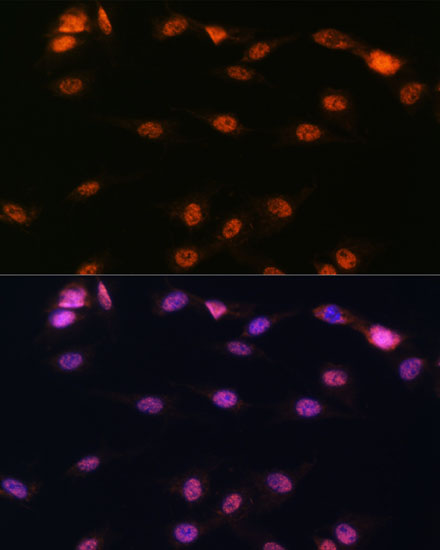
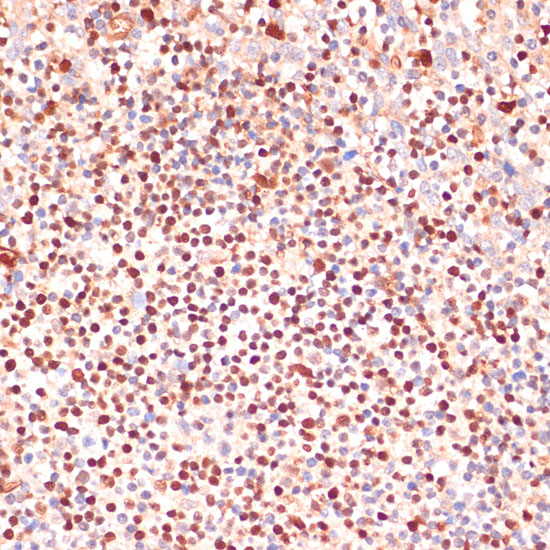
5-Lipoxygenase (5-LO, ALOX5) is an important catalytic enzyme responsible for the biosynthesis of leukotriene LTA4 from arachidonic acid (1,2). Leukotriene synthesis also requires 5-lipoxygenase-activating protein (FLAP, ALOX5AP), a nuclear membrane-bound protein that binds arachidonic acid and is thought to activate 5-LO. A number of related leukotrienes (i.e. B4, C4, D4) are derived from LTA4 and together these lipid mediators function in immune reaction regulation. 5-LO is primarily expressed in polymorphonuclear leukocytes, peripheral blood monocytes, macrophages, and mast cells (1,3). Overexpression of 5-LO protein is seen in certain cancer cells and is associated with poor diagnosis (1,4). Depending upon the cell type, 5-LO is localized to either the cytosol or the nucleus of quiescent cells (5). Following stimulation, 5-LO translocates to the nucleus and associates with FLAP to catalyze LTA4 synthesis (2,3). Phosphorylation of specific residues can regulate 5-LO enzymatic activity. Phosphorylation of 5-LO at Ser523 by PKA family kinases inhibits oxygenase activity (6,7) while MAPKAP2 and ERK family kinase phosphorylation at Ser271 and Ser663 stimulates 5-LO enzymatic activity in vivo (8,9).