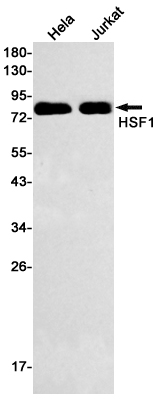
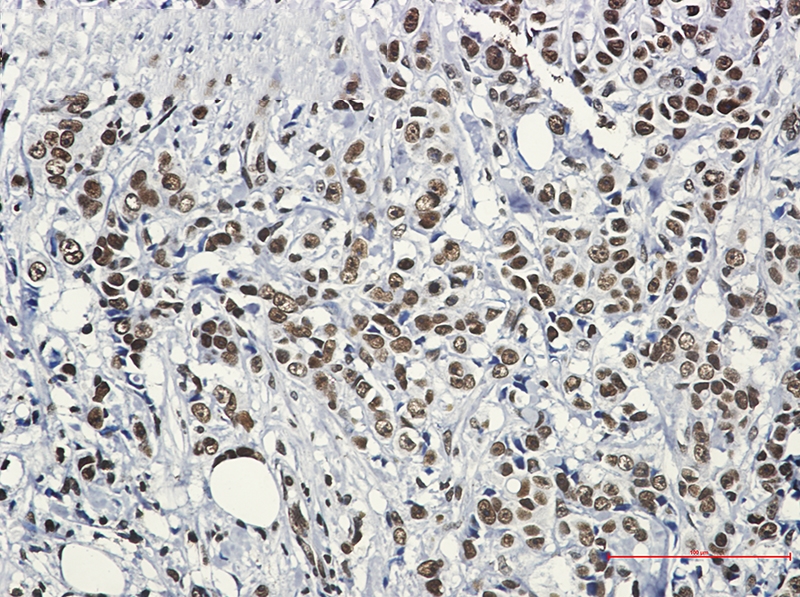
Swiss-Prot Acc.Q00613.Function as a stress-inducible and DNA-binding transcription factor that plays a central role in the transcriptional activation of the heat shock response (HSR), leading to the expression of a large class of molecular chaperones heat shock proteins (HSPs) that protect cells from cellular insults' damage (PubMed:1871105, PubMed:11447121, PubMed:1986252, PubMed:7760831, PubMed:7623826, PubMed:8946918, PubMed:8940068, PubMed:9341107, PubMed:9121459, PubMed:9727490, PubMed:9499401, PubMed:9535852, PubMed:12659875, PubMed:12917326, PubMed:15016915, PubMed:25963659, PubMed:26754925). In unstressed cells, is present in a HSP90-containing multichaperone complex that maintains it in a non-DNA-binding inactivated monomeric form (PubMed:9727490, PubMed:11583998, PubMed:16278218). Upon exposure to heat and other stress stimuli, undergoes homotrimerization and activates HSP gene transcription through binding to site-specific heat shock elements (HSEs) present in the promoter regions of HSP genes (PubMed:1871105, PubMed:1986252, PubMed:8455624, PubMed:7935471, PubMed:7623826, PubMed:8940068, PubMed:9727490, PubMed:9499401, PubMed:10359787, PubMed:11583998, PubMed:12659875, PubMed:16278218, PubMed:25963659, PubMed:26754925). Activation is reversible, and during the attenuation and recovery phase period of the HSR, returns to its unactivated form (PubMed:11583998, PubMed:16278218). Binds to inverted 5'-NGAAN-3' pentamer DNA sequences (PubMed:1986252, PubMed:26727489). Binds to chromatin at heat shock gene promoters (PubMed:25963659). Plays also several other functions independently of its transcriptional activity. Involved in the repression of Ras-induced transcriptional activation of the c-fos gene in heat-stressed cells (PubMed:9341107). Positively regulates pre-mRNA 3'-end processing and polyadenylation of HSP70 mRNA upon heat-stressed cells in a symplekin (SYMPK)-dependent manner (PubMed:14707147). Plays a role in nuclear export of stress-induced HSP70 mRNA (PubMed:17897941). Plays a role in the regulation of mitotic progression (PubMed:18794143). Plays also a role as a negative regulator of non-homologous end joining (NHEJ) repair activity in a DNA damage-dependent manner (PubMed:26359349). Involved in stress-induced cancer cell proliferation in a IER5-dependent manner (PubMed:26754925).