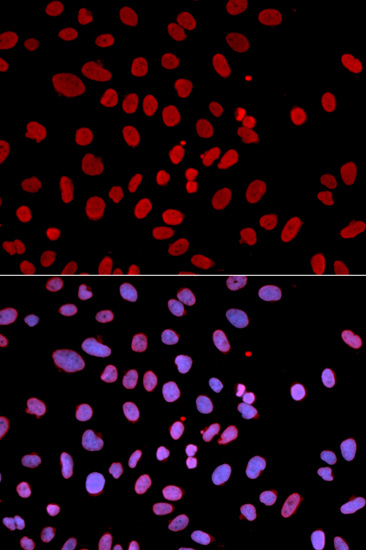
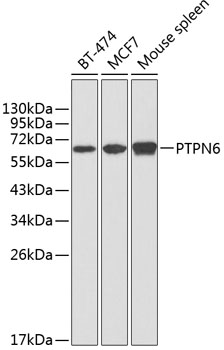
SHP-1 (PTPN6) is a non-receptor protein tyrosine phosphatase that is expressed primarily in hematopoietic cells. The enzyme is composed of two SH2 domains, a tyrosine phosphatase catalytic domain, and a carboxy-terminal regulatory domain (1). SHP-1 removes phosphates from target proteins to downregulate several tyrosine kinase-regulated pathways. In hematopoietic cells, the amino-terminal SH2 domain of SHP-1 binds to tyrosine phosphorylated erythropoietin receptors (EpoR) to negatively regulate hematopoietic growth (2). Overexpression of SHP-1 in epithelial cells results in dephosphorylation of the Ros receptor tyrosine kinase and subsequent downregulation of Ros-dependent cell proliferation and transformation (3). Following ligand binding in myeloid cells, SHP-1 associates with the IL-3R β chain and downregulates IL-3-induced tyrosine phosphorylation and cell proliferation (4). Because SHP-1 downregulates various proliferation pathways, SHP-1 is considered a potential tumor suppressor and angiogenesis regulator (5,6).