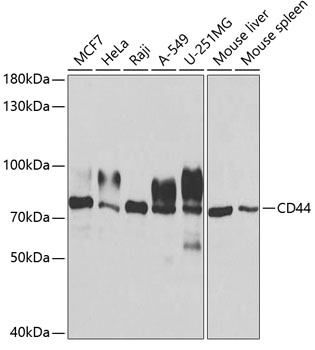
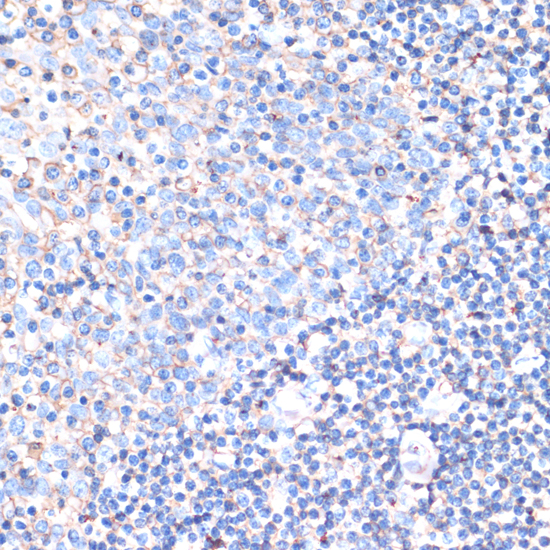
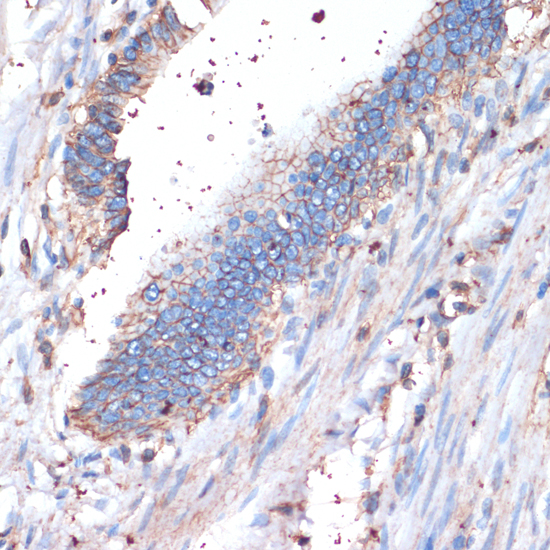
CD44 is a type I transmembrane glycoprotein that mediates cell-cell and cell-matrix interaction through its affinity for hyaluronic acid (HA) and possibly through other parts of the extracellular matrix (ECM). CD44 is highly polymorphic, possesses a number of alternative splice variants and undergoes extensive post-translational modifications (1,2). Increased surface levels of CD44 are characteristic of T cell activation, and expression of the protein is upregulated during the inflammatory response. Interactions between CD44 and HER2 have been linked to an increase in ovarian carcinoma cell growth (1-3). CD44 interacts with ezrin, radixin and moesin (ERM), linking the actin cytoskeleton to the plasma membrane and the ECM (4-6). CD44 is constitutively phosphorylated at Ser325 in resting cells. Activation of PKC results in phosphorylation of Ser291, dephosphorylation of Ser325, disassociation of ezrin from CD44, and directional motility (4).